Capable of accurately measuring oxygen at very low levels, optical oxygen sensors are an effective and affordable alternative to the traditional electrochemical sensors. They are based on the principle of fluorescence quenching by oxygen and rely on the light energy measurement versus chemical reaction measurement, which allows for a vast array of functional and technological benefits.
That is why optical sensors are quickly becoming the popular go-to solution in many fields where oxygen measurement is a key parameter. In the last decade, these sensors have been widely utilized in the lab, process environments and field measurement applications.
In this article, I would like to give a brief overview of the two alternative technologies with the goal of illustrating the advantages that optical oxygen sensors offer in comparison with their electrochemical counterparts.
How do the two technologies work?
In an electrochemical sensor, both galvanic and polarographic, oxygen permeates through the sensor membrane and undergoes a chemical reduction reaction, which produces an electrical signal that is proportional to the concentration of oxygen in the sample. During this type of measurement, oxygen gets consumed in the process, which means that a constant inflow of sample at a certain speed is required to maintain measurement accuracy.
On the other hand, no oxygen consumption occurs in the optical sensor. Here, the polymer membrane contains a special luminescent dye that glows bright (thick pink halo in the image below) when exposed to a light source in the absence of oxygen. When oxygen molecules are present in the sample, they collide with the dye molecules and quench their luminescence (reduced pink halo in the image below) before transmitting back to the detector. This effect is called “quenching.” The more oxygen in the sample, the less light is getting back. The difference being the indicator of oxygen concentration in the sample. Optical technology is not flow dependent, making the measurement process easier and faster.
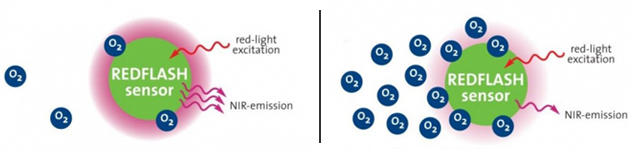
Functional benefits:
Because the electrochemical sensors consist of electrodes and require polarization to operate, certain types need several minutes of warm-up time before testing can begin. In comparison, there is no such downtime with optical sensors. They readily perform detection upon start, providing unprecedentedly low response times from a few seconds down to fractions of a second in special cases.
Thanks to the versatile design of optical technology, sensors can easily be miniaturized, opening opportunities to many applications not attainable using electrochemical sensors. The possibility of contactless and long-term field measurements are other unique attributes of optical sensors.
Additionally, optical oxygen sensors tend to be more accurate than their electrochemical peers since they are not affected by hydrogen sulfide or other gasses that can permeate an electrochemical membrane.
What kind of maintenance is required?
There are quite a few components that require periodic maintenance in electrochemical sensors. The electrolyte solution that provides electrical conductivity needs refilled periodically, sensor membranes wear out requiring replacement, and electrodes oxidize needing cleaning. In addition, electrochemical sensors must be calibrated before every use, whereas optical sensors can implement long-term in-situ testing.
With optical sensors, the only cause of maintenance stems from the luminescence dye potentially bleaching out and degrading with time. When the signal intensity indicator drops below a certain point, a sensor replacement is required. In our experience, the optical sensors can last years, depending on usage.
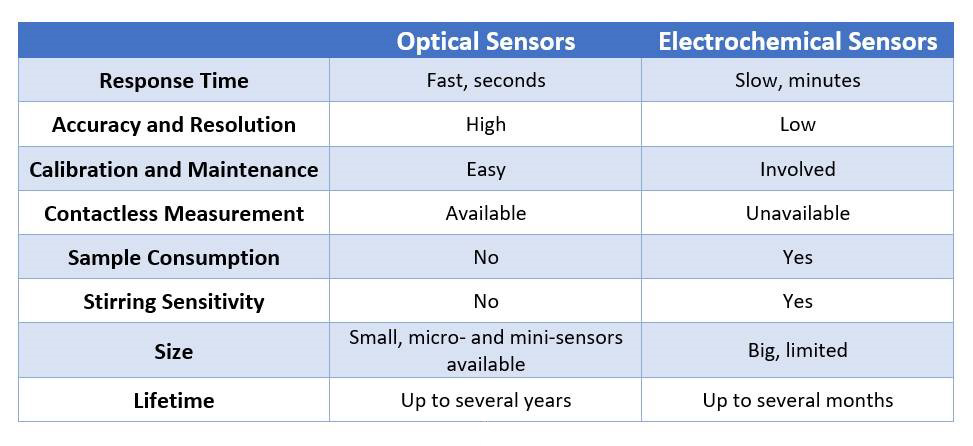
To summarize why you should trust optical sensors over electrochemical:
For more information on the available optical oxygen sensors, contact Nadia Savelyeva at nadia.savelyeva@ohiolumex.com.
